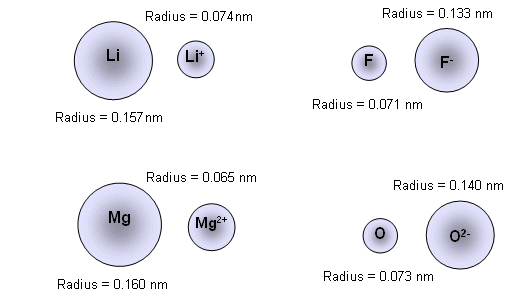
Ionic Radius Trends, Basic Introduction, Periodic Table, Sizes of Isoelectric Ions, Chemistry - YouTube
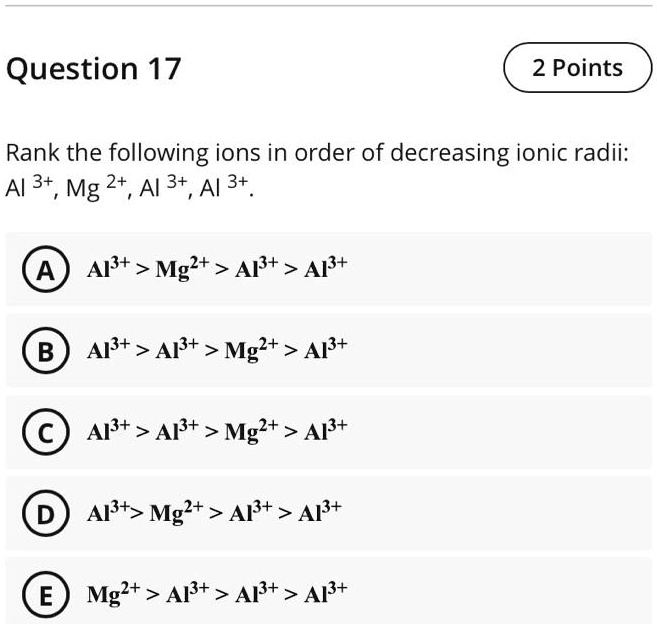
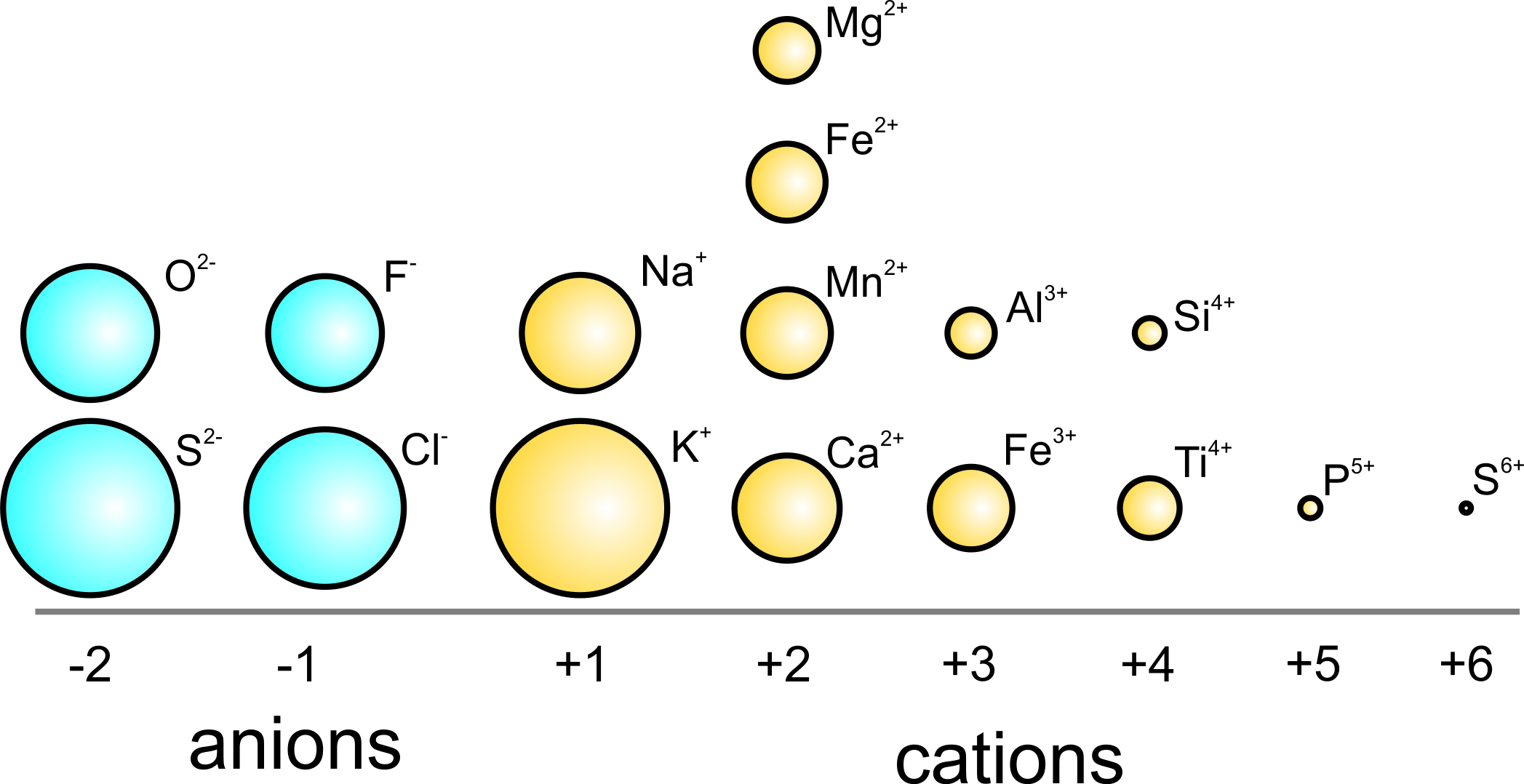
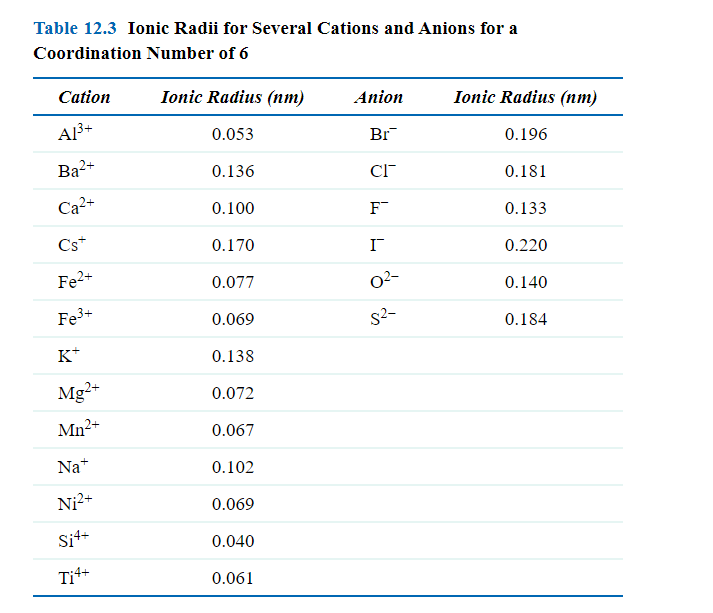
SOLVED: Question 17 2 Points Rank the following ions in order of decreasing ionic radii: Al 3+, Mg 2+, Al 3+,Al 3+ Al3+ > Mg2+ > Ap+ > Ap+ AP+ AP+ Mg2+ >

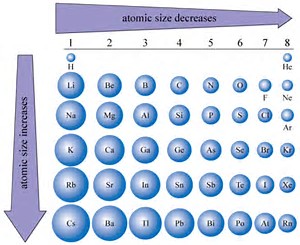
Size of Atoms - Trends Atomic Radii Li Na K Rb Cs Cl S P Si Al Br Se As Ge Ga I Te SbSn In Tl Pb Bi Mg Ca Sr

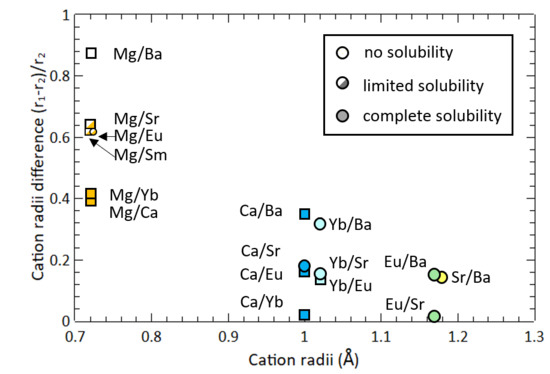
Mean ionic radii of tetrahedral and octahedral sites (r A , r B ) of Ni... | Download Scientific Diagram
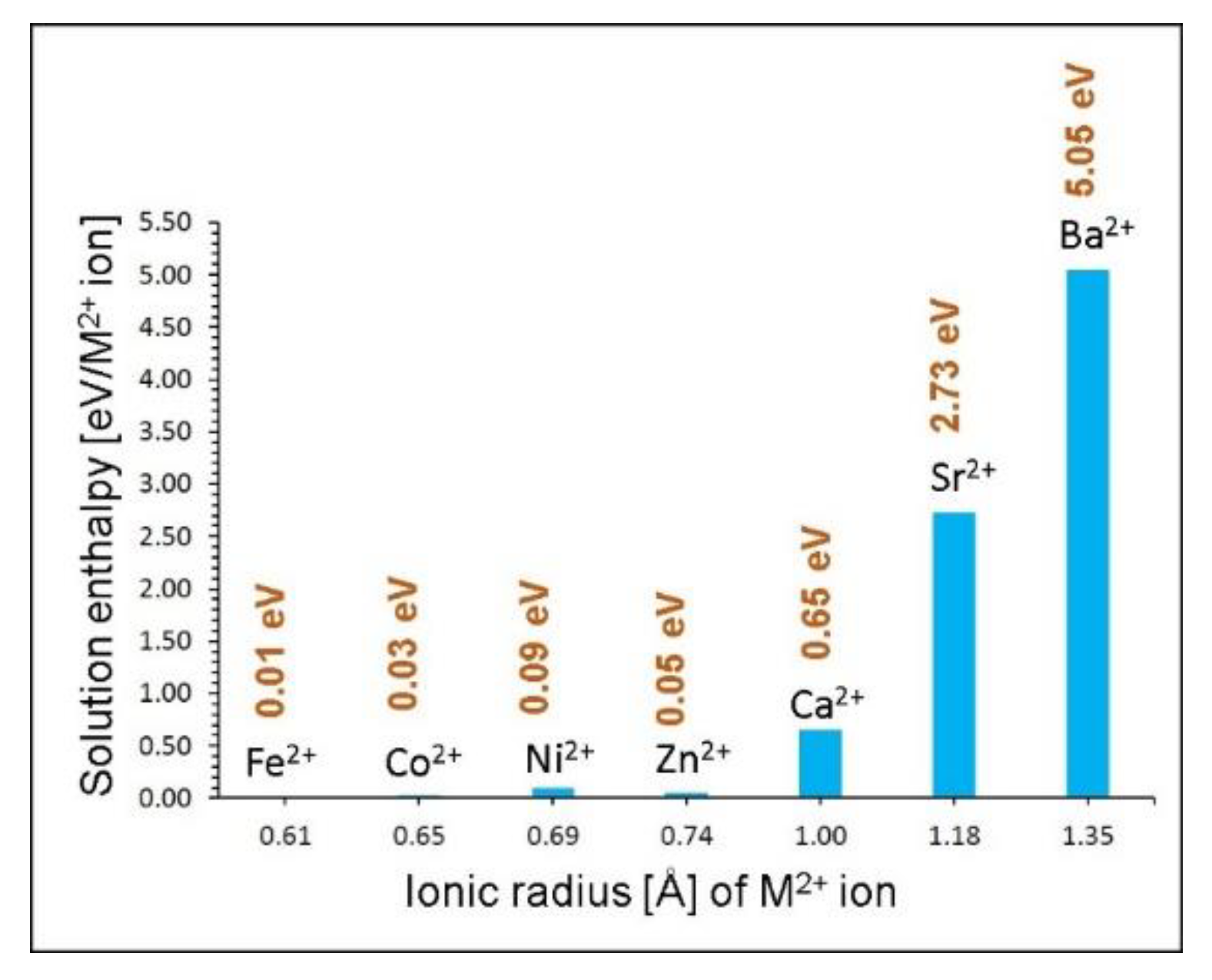
Energies | Free Full-Text | Mg6MnO8 as a Magnesium-Ion Battery Material: Defects, Dopants and Mg-Ion Transport
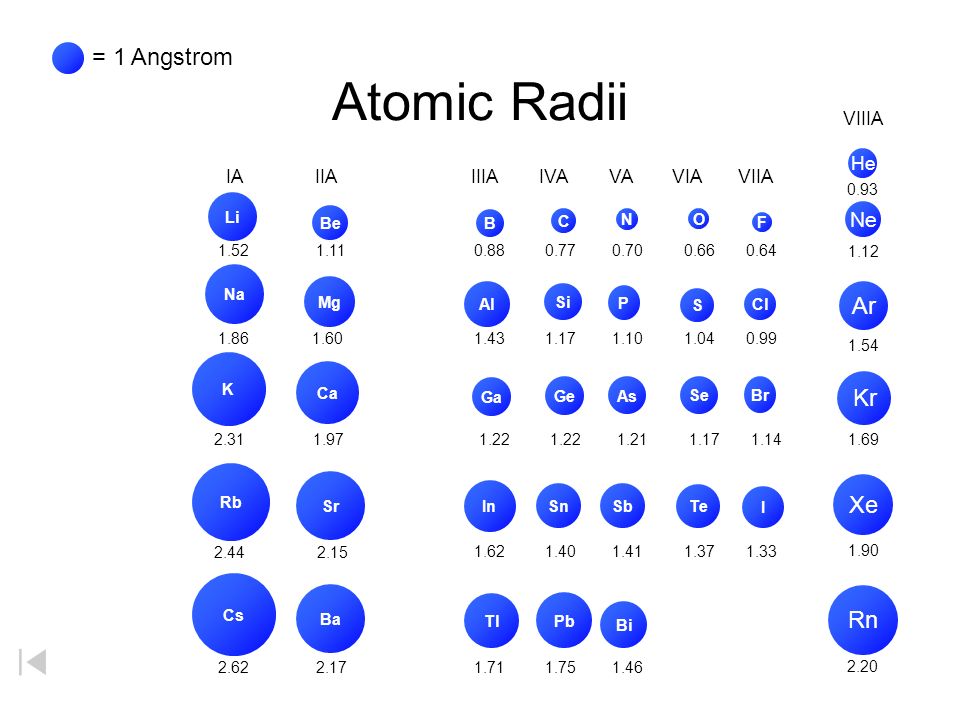
Size of Atoms - Trends Atomic Radii Li Na K Rb Cs Cl S P Si Al Br Se As Ge Ga I Te SbSn In Tl Pb Bi Mg Ca Sr

Ion Magnesium carbonate Chemical compound Inorganic compound, information symbol, text, trademark png | PNGEgg